NIH3T3 Transfection Information
NIH3T3 cells (also known as 3T3 cells) are a standard fibroblast cell line utilized in biological research. The 3T3 cell line was originally established from a Swiss albino mouse embryo tissue in 1962 by two scientists at New York University School of Medicine. ‘3T3’ is an abbreviation from the original label of “3-day transfer, inoculum 3 x 105 cells”, which refers to the protocol used to culture the cell line from primary mouse embryonic fibroblast cells. Utilizing the 3T3 protocol, the researchers at NYU were able induce spontaneous immortalization in the cells after 20-30 generations in culture.
Research Using Transfected NIH3T3 Cells
- 3T3 is useful in the cultivation of keratinocytesas keratinocytes benefit from exposure to 3T3 growth factors.
- 3T3 cells are well suited for transfection studies using DNA. Although this original cell line was subject to multiple cycles of subcloning before it was receptive to transformations, 3T3 is capable of undergoing spontaneous transformations in culture.
- Transformations using 3T3 are often performed with DNA from tumorigenic cells in order to study and demonstrate the presence of transforming oncogenes.
NIH3T3 Transfection Protocol
An optimized NIH3T3 Transfection Kit is available from Altogen Biosystems, which includes:
- NIH3T3 Transfection Reagent (0.5 ml / 1.5 ml / 8.0 ml)
- Transfection Enhancer (0.5 ml), and
- Complex Condenser (0.5 ml)
This lipid-based transfection reagent is optimized for transfection of DNA and RNA into NIH3T3 cells following either a forward or reverse protocol. The protocol for a 24-well plate to transfect NIH3T3 cells is as follows:
- Plate 10,000-15,000 NIH3T3 cells per well in 0.5 ml of complete growth medium 12-24 hours prior to transfection
- Wash with 1xPBS and add 0.5 ml of fresh growth medium
- Prepare transfection complexes by mixing 40 µl of serum-free medium, 5.5 µl of transfection reagent, and (referred to a final volume including growth medium)
- 750 ng DNA (or mRNA), or
- 30 nM – 50 nM of siRNA (or microRNA)
- Incubate transfection complexes at RT for 15-30 minutes
- Optional: Add 2 µl of Complex Condenser. This reagent increases transfection efficiency by reducing the size of transfection complex; however, it may increase cell toxicity
- Add prepared transfection complexes to 0.5 ml of complete growth medium with NIH3T3 cells (from step 2)
- Incubate cells at 37ºC in a humidified CO2incubator
- Assay for phenotype or target gene expression 48-72 hours after transfection
Optional: Adding Transfection Enhancer reagent can increase transfection efficiency. Add 2 µl of Transfection Enhancer reagent 12-24 hours after transfection
If the viability of NIH3T3 cells being transfected is affected at 16-24 hours post-transfection, changing the growth medium and eliminating redundant exposure of cells to transfectant can decrease the level of cytotoxicity
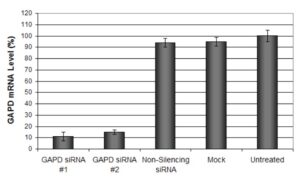
Figure: GAPDH mRNA levels were quantified using qPCR in NIH3T3 cells transfected with siRNAs targeting GAPDH or non-silencing siRNA. 48 hours post-transfection, the cells were harvested and analyzed by qRT-PCR for GAPDH mRNA expression levels. Data were normalized to the 18S rRNA signal and expressed as relative to the untreated samples. Data are means ± SD (n=3).
NIH3T3 Transfection Efficiency
Transfection is a method to integrate protein, RNA and DNA molecules into cultured cells. Efficiency of transfection is a phrase used to sum up the success of an experiment which contains many variables. A successful transfection depends upon the optimization of these:
- cell density
- cell type
- transfection reagent
- test article concentration
- media components used (i.e. serum, medium, antibiotics)
Transient transfection gene knockdown can exhibit positive results as early as 4 hours post-transfection, with typical maximal effects at 24-48 hours and compete loss of the transient effect at 96 hours. However, the analysis of transfected plasmid DNA needs to be altered to allow DNA uptake, expression of the plasmid and efficacy of the exogenous target gene.
Links
List of Companies Offering Lab Services
NIH3T3 Cell Transfection Resource
Transfection: Cells and Molecular Biology Research Methods, Protocols and Lab Techniques
NIH3T3 Cells │ NIH3T3 Cell Culture │ NIH3T3 Transfection │ Get NIH 3T3 Transfection Reagent